 Hydrogen is the only atom for which the Schrödinger equation has an exact solution.In compounds, hydrogen can take a negative charge (H -) or a positive charge (H +).Hydrogen is also used in fuel cells that react hydrogen and oxygen to produce water and electricity. It is gaining importance as an alternate fuel that produces energy by combustion, similar to what happens in fossil fuel engines. Hydrogen has many uses, though most hydrogen is used for processing fossil fuels and in the production of ammonia.Solid crystalline metallic hydrogen has the lowest density of any crystalline solid. The gas and liquid are nonmetals, but when hydrogen is compressed into a solid, the element is an alkali metal. The physical form of hydrogen at room temperature and pressure is a colorless and odorless gas.Hydrogen may be produced by reacting metals with acids (e.g., zinc with hydrochloric acid).Hydrogen compounds commonly are called hydrides.However, it's an oxidizer, which is why hydrogen is so explosive in air or with oxygen. While many people consider oxygen to be flammable, it actually doesn't burn. It is used as a fuel by the space shuttle main engine and was associated with the famous explosion of the Hindenburg airship. Tritium is the isotope with 1 proton, 2 neutrons, and 1 electron. Although this isotope is heavier than protium, deuterium is not radioactive. This makes hydrogen the only element that can have atoms without any neutrons! Deuterium has 1 proton, 1 neutron, and 1 electron. The most common isotope of hydrogen is protium, which has 1 proton, 0 neutrons, and 1 electron. There are three natural isotopes of hydrogen: protium, deuterium, and tritium.The element remains common at the Earth's surface, where it is bound into water and hydrocarbons to be the third most abundant element. Hydrogen exists as a pure element on Earth as a diatomic gas, H 2, but it's rare in Earth's atmosphere because it is light enough to escape gravity and bleed into space. Although hydrogen is the most abundant element in the human body in terms of numbers of atoms of the element, it's only 3rd in abundance by mass, after oxygen and carbon, because hydrogen is so light. 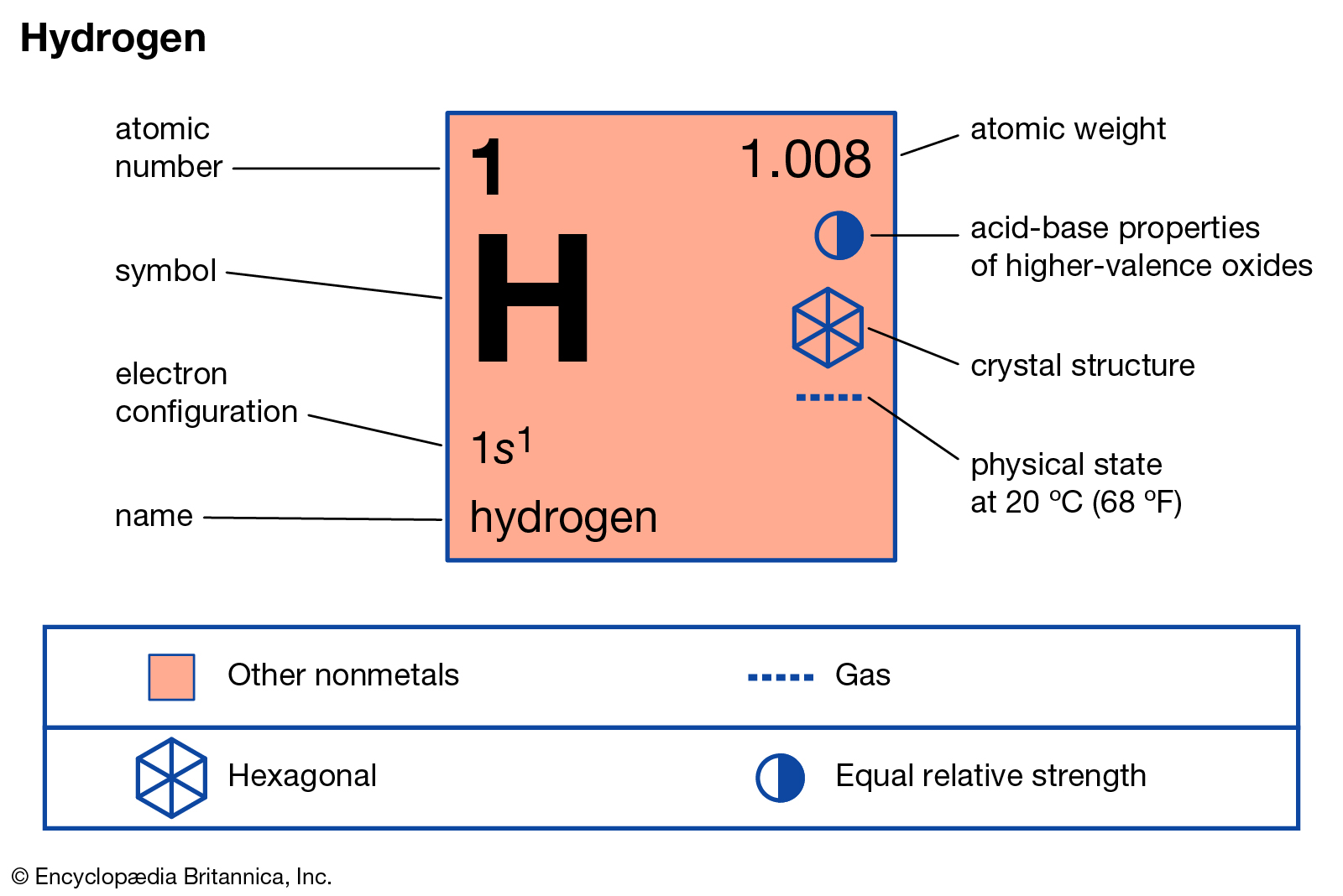
About 90% of the atoms and 75% of the element mass of the universe is hydrogen, usually in the atomic state or as plasma. Hydrogen is the most abundant element.Join Seneca to get 250+ free exam board specfic A Level, GCSE, KS3 & KS2 online courses. The denominator is 'the sum of all the isotope abundances'. The numerator is 'the sum of the isotope abundance times the isotope mass'. It takes into account how often each isotope is found (the isotope abundance). The relative atomic mass (Ar) is the average mass of all of the isotopes of an element. It is used in thermonuclear fusion weapons. Other atoms typically do not have round-number atomic masses. Carbon-12, an atom of carbon-containing six neutrons, has an atomic mass of 12 amu. A single atom’s atomic mass is just its total mass, and it is commonly given in atomic mass units or amu. An isotope is named after the element and the mass number of its atoms. The atomic mass of an atom is a feature that is closely related to its mass number. Tritium is a hydrogen atom with 1 proton and 2 neutrons. Hydrogen-1 is the most abundant (most common) isotope of hydrogen. 
Around 0.02% of hydrogen atoms are deuterium. Deuterium is a hydrogen atom with 1 proton and 1 neutron.It is used in hydrogen fuel cells and the production of plastics. Protium is a hydrogen atom with 1 proton and 0 neutrons.But atoms of the same element can have different numbers of neutrons: these atoms are known as isotopes. 
Atoms of the same element always have the same number of protons (or have the same atomic number). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
